The context
Kidney transplantation is currently the best therapeutic option with around 1,500,000 patients living with a kidney transplant. However, long-term graft survival has not been improved in the past 15 years and rejection is a major cause of graft loss, representing at least 12 billion € extra costs per year.
There is a major unmet need: a lack of robust tools to stratify the risk of rejection, graft function decline and graft loss.
To tackle this issue, the EU-TRAIN project aims at developing a risk stratification system in kidney transplantation by integrating multidimensional data from patients’ follow-up.
An innovative and
Multidimensional approach
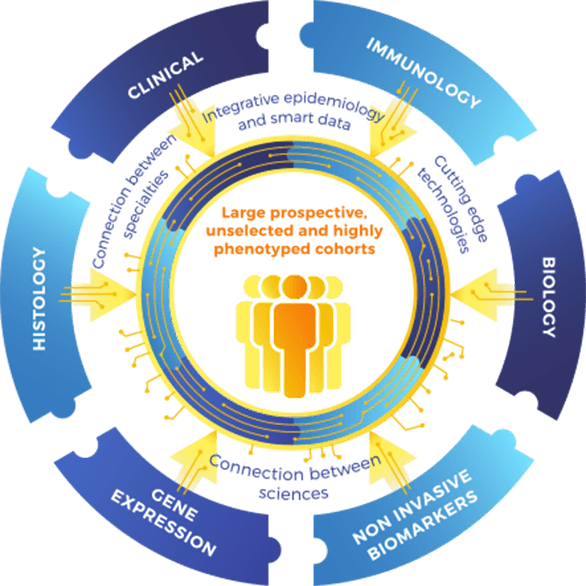
The traditional approach of patient’s follow-up in kidney transplantation bears several limitations. There is a lack of connection between the different specialties revolving around the kidney transplant recipient (External circle, Figure 1).
Moreover, approaches used to monitor immune-mediated allograft damage are nonspecific markers such as serum creatinine level and proteinuria, and the current diagnostic system exclusively relies on histopathology examination of surveillance biopsies, which is the actual gold standard. Histopathology is not sufficient by itself for precision diagnostics and not necessarily associated with outcome prediction. It lacks specificity and sensitivity and provides no etiopathology of the underlying processes, leading to incorrect diagnoses.
The EU-TRAIN consortium proposes an integrative and multidimensional approach connecting the dots between Omics (gene expression analysis), allograft phenotypes (tissue specific injury), immunological biomarkers, epidemiology, clinical data and therapeutics (Inner circle, Figure 1). Unlike other initiatives, EU-TRAIN will not focus on a specific biomarker but will define a biomarker ensemble (“multimarker approach”), driven from existing large cohorts (reference sets) gathered over the last decade by partners of the consortium, and the large prospective and highly phenotyped cohort of transplant patients from the EU-TRAIN study (Population inside the circle, Figure 1).
Eu-tracer
Our methodology
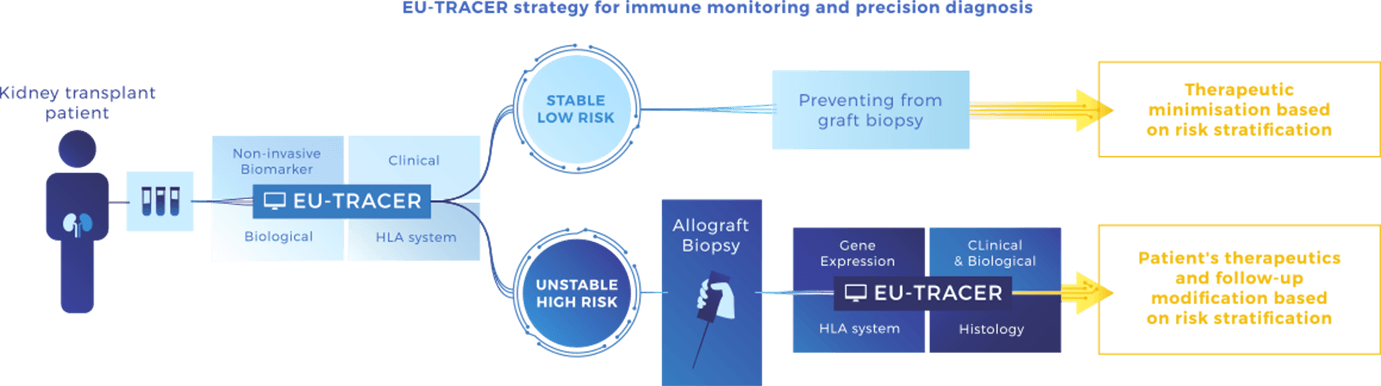
We have designed EU-TRAIN to cover the 2 major clinical situations of transplant patients:
1. Low risk (stable): defined by non-invasive tests (blood) to monitor patients and prevent unnecessary invasive biopsies and their costly procedure.
2. High risk (unstable): based on allograft assessment including gene expression profiling. We will develop a risk stratification system integrating a molecular and a conventional assessment that will estimate more accurately the true disease activity, stage and response to therapy.
Multidimensional data including non-invasive and invasive biomarkers from the EU-TRAIN cohort will be gathered in a prediction system called EU-TRACER, that will allow early stratification in terms of individual risk for allograft rejection as defined above, response to standard of care and emerging therapies, and allograft loss prediction.
How will EU-TRACER work?
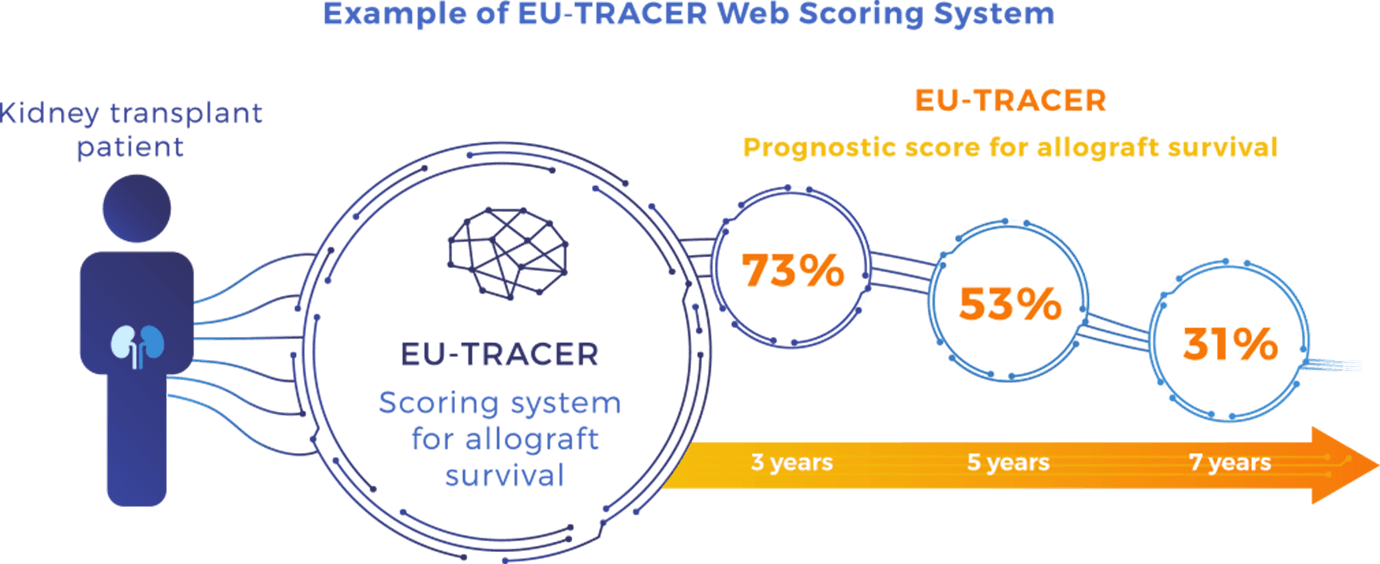
For each patient, a EU-TRACER score will be calculated by entering the patient’s medical parameters of interest on a web interface system. This will be achieved by adapted prognostication algorithms that will be fed by EU-TRAIN patients’ data in a dynamic way. The results will be displayed by showing the patient’s kidney allograft survival probability at 3, 5 and 7-year post-transplant depending on the EU-TRACER score.
The EU-TRACER score will be calculated using information from reference set cohorts and EU-TRAIN prospective cohorts :
- - Status at the time of transplantation: donor and recipient characteristics, transplant characteristics and donor and recipient immunological data comprising 85 parameters. Among those, sex and gender are taken into account from both donor and recipient where it plays a central role because of distinct allograft mass supply, differential level of sensitisation, different rejection profiles and distinct vulnerability to injuries. This critical point has not been addressed so far in transplantation despite the fact that sex and gender have cumulative and potential strong interactions.
- - Routine biology lab tests for drug monitoring, viral load, allograft function parameters, treatment and medical complication record (cancers, infections, cardiovascular diseases);
- - Prospective allograft phenotyping on kidney transplant biopsies graded according to international Banff classification, centralised and standardised by virtual microscopy;
- - Kidney allograft gene expression on transplant biopsies;
- - Prospective HLA, non-HLA and other endothelial target antibody monitoring using sensitive single antigen technologies for antibody screening and characterisation;
- - New immunological biomarkers: cell-based functional assays and peripheral (blood, urine transcriptomics;
- - Outcome parameters comprising allograft rejection, kidney allograft loss and patient death.
Expected results
Based on previous studies on reference sets, we have evaluated that incorporating relevant non-invasive biomarkers and gene expression data to the actual standard of care parameters, has the potential to improve the rejection reclassification by 25% and the overall risk of rejection and allograft loss stratification accuracy by 30%. The EU-TRAIN consortium decision will be made based on a careful selection of the most predictive and informative biomarkers that provide independent and significant improvement of patient risk stratification.
Milestones
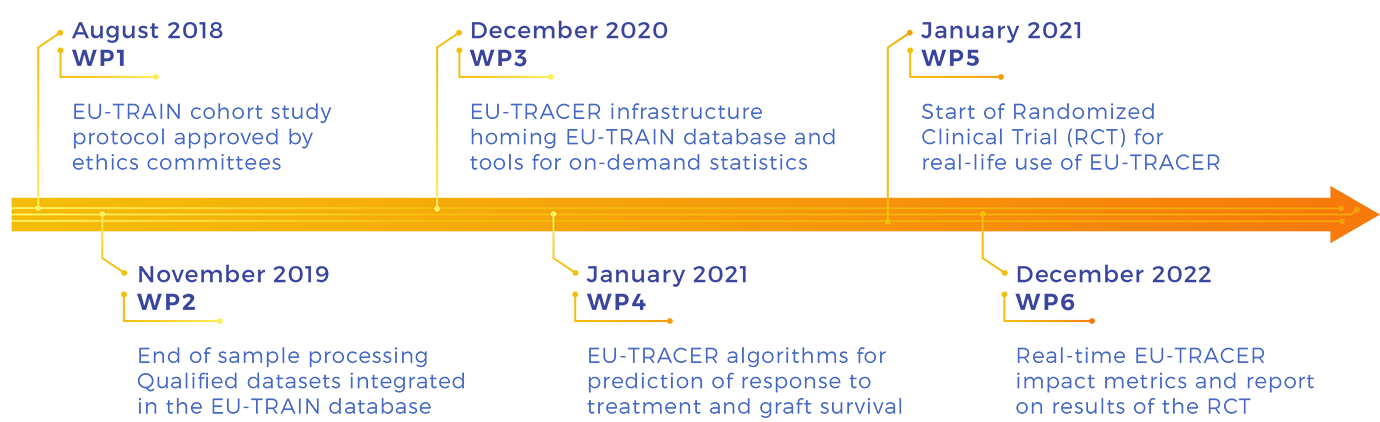
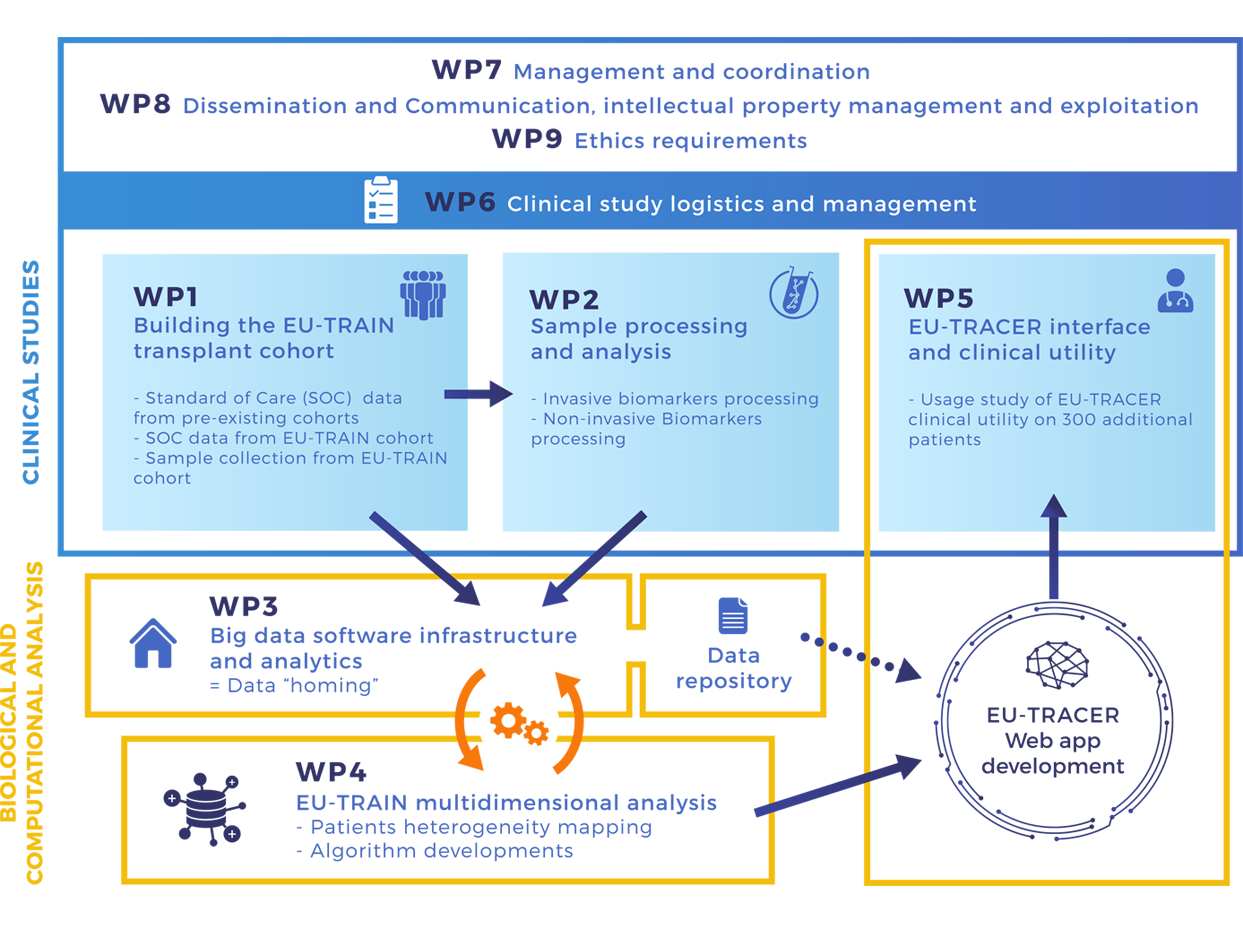
Workpackages



 This project has received funding from the European
This project has received funding from the European
